 
Copper is an element.Įlements are substances which can't be split into anything simpler by chemical means. Again copper is a simpler substance than copper(II) oxide.īut there is nothing you can do chemically to turn copper into anything still simpler. If you pass hydrogen over hot copper(II) oxide, the hydrogen removes the oxygen, and leaves pinkish-brown copper, Cu. Copper(II) oxide is obviously a simpler substance than copper(II) nitrate. If you heat some blue copper(II) nitrate crystals, Cu(NO 3) 2,3H 2O, you get a chemical change and it turns into black copper(II) oxide, CuO, giving off nitrogen dioxide, oxygen and steam in the process. Cu is the symbol for copper the others are fairly obvious. They are just there to illustrate the complexity of various substances. Don't worry about it if you haven't done any work on symbols yet. I am going to include some chemical formulae in what comes next. It also takes a quick look at the Periodic Table - a really important tool in chemistry. With a bit of trial and error, you'll be able to determine whether hot molecules of water move more quickly than cold ones.This page explains the difference between an element and a compound, and how both of these differ from mixtures. You'll need some cold water, hot water, food coloring, and an eye dropper. Jump online to check out the Water Molecules on the Move experiment.You'll need just a few simple materials and help from a friend or family member. To experiment with some different types of molecules that don't mix together, try the fun Mixing Oil and Water experiment. Do molecules bond together? Sometimes they do…and sometimes they don't! It depends upon what they're made of.To enjoy that glass of water molecules to the fullest, you should add some more water molecules to it…except add some solid ones this time! Reach into the freezer and grab a few ice cubes, otherwise known as frozen water molecules! Before you drink your water, spend some time looking at it closely, comparing the solid and liquid water molecules and the differences in their appearance. Grab a glass and turn on the tap and fill your glass with a bunch of water molecules. Do molecules always look the same? Not necessarily! You have a source of molecules in your kitchen. 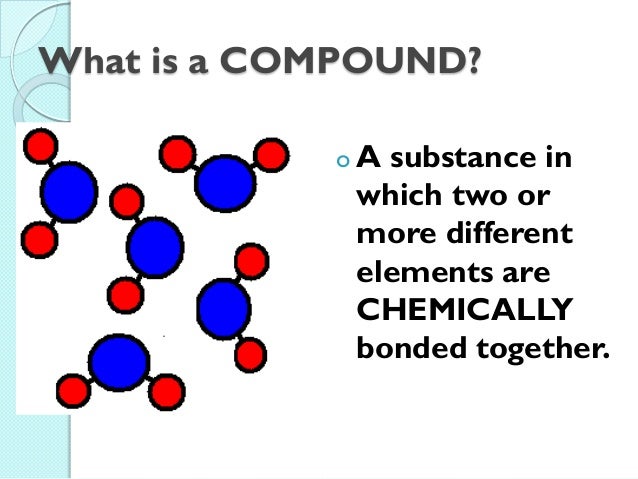
An example of a compound formed with ionic bonds is sodium chloride (NaCl), otherwise known as table salt!Īre you ready to experiment with molecules? Be sure to explore the following activities with a friend or family member: In an ionic bond, electrons get transferred from the metal to the nonmetal substance. Ionic bonds form between a metal and a nonmetal substance. This is what makes metals good conductors of heat and electricity. The valence atoms in metals move freely and thus form bonds easily. The bonds between the two hydrogen atoms and the oxygen atom in a molecule of water are covalent bonds.Īs its name implies, a metallic bond occurs between metallic substances. In a covalent bond, electrons are shared between atoms. However, there are three primary types of chemical bonds that you'll see most often: covalent, metallic, and ionic.Ĭovalent bonds form between nonmetallic materials. A common compound everyone is familiar with results from the chemical bonding of two atoms of hydrogen with one atom of oxygen to form a molecule of water (H 2O).Ītoms can chemically bond in many ways. If a molecule forms from atoms of two or more different elements, we call it a compound. For example, when three oxygen atoms bond together, they form a molecule of ozone (O 3). Sometimes the atoms are all from the same element. 
When two or more atoms chemically bond together, they form a molecule. This attraction results from the properties and characteristics of the atoms' outermost electrons, which are known as valence electrons. Why exactly do atoms tend to combine together? Why aren't they happy just hanging out by themselves? As it turns out, it's purely a matter of chemistry.Ītoms form chemical bonds with other atoms when there's an electrostatic attraction between them. 
Likewise, they make up all the matter around us, from mountains and seas to planets and stars. Too small to see, those atoms combine in an infinite number of ways to make us who and what we are. On the smallest scale, we are made up of countless tiny atoms. And yet all of these things are composed of variations of the same small set of materials we know as elements. Over seven billion human beings and an untold number of animals inhabit Earth. Here on Earth, we have tall mountains, deep seas, and miles upon miles of all sorts of land. We have the planets and the Sun in our solar system, which is part of the larger Milky Way galaxy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
